Doing business in the Middle East and Africa can be attractive, but challenging
In a Voice of the Industry Market Outlook survey of more than 6,000 healthcare decision-makers worldwide, respondents named Africa and the GCC respectively as the two most important markets for their business. Yet despite their attractiveness, these markets pose challenges that differ by country.
There is a tendency, of course, to lump together the markets of the Middle East and indeed Africa under a convenient umbrella term, such as MEA or MENA. But as with other regions worldwide, the reality of operating within these is more complex, owing to different factors.
In spatial terms alone, Africa is far bigger than the United States, Canada, China and western Europe combined, while there is more to the Middle East than shining metropolises such as Dubai, Riyadh and Doha.
Regulations, certification and collecting payments keep decision-makers awake at night
Correspondingly, the survey showed that barriers including regulations and certifications, tariffs/import duties, customs clearance, logistics and collecting payments all varied in importance across markets in the Middle East and Africa.
Take for example the GCC, where regulations and certification were identified by the survey participants as the two biggest problems when doing business.
Knowing how to navigate these complexities can be a head-scratcher, but where better to begin than consulting outsourcing experts on the ground, who can save a lot of worry (and money)?
We spoke with one such company, Pedigri Technologies, who revealed how they overcome challenges typically faced by international manufacturers in the Middle East and Africa.
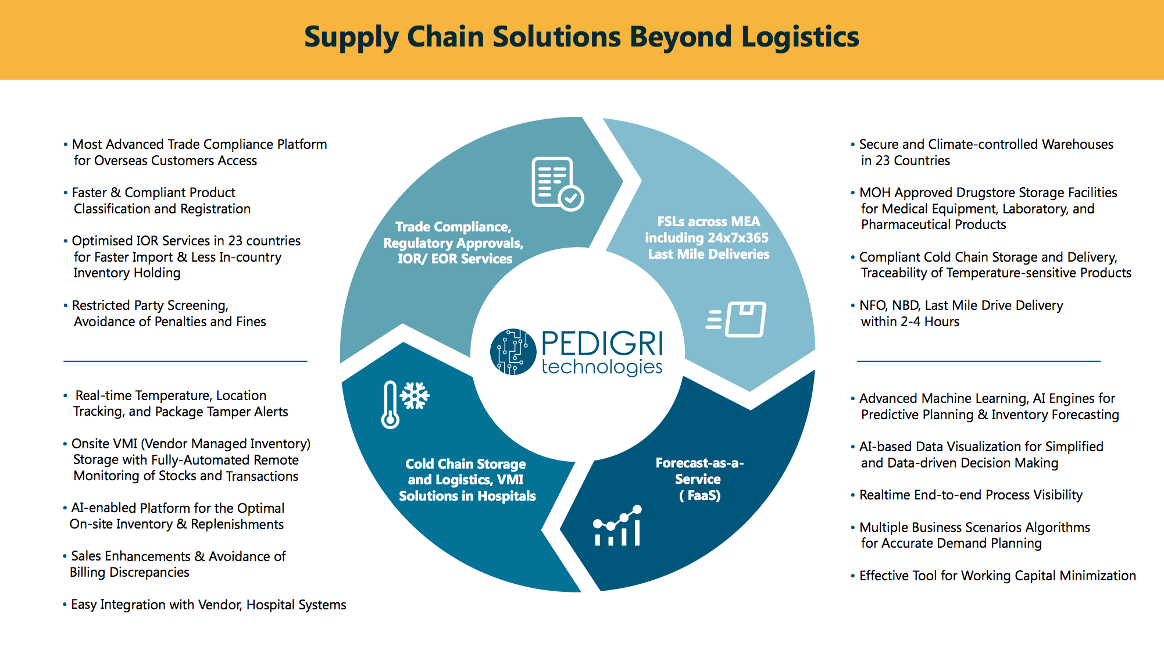
Headquartered in Dubai, and established for two decades, the company is ideally placed to help international manufacturers with their regulatory, customs clearance and last mile delivery needs.
Painless outsourcing
Pedigri Technologies sees itself as a local solutions partner. Its business consists of more than 200 dedicated professionals whose countries of origin are the markets it serves. This means that it is innately familiar with the cultural nuances, languages and trends of the territories it operates in.
But it's not just the human touch that can make a difference. It also makes the most of the latest digital capabilities, such as Artificial Intelligence, Machine Learning, Robotic Process Automation, Real-time Monitoring and Big Data.
These strengths combined ultimately save time, money and complexities, meaning that wherever you are in the world, you can enjoy a good night's sleep knowing that your supply chain and inventory needs are taken care of.
Faster import solutions
Delays in the import of medical equipment and spare parts into countries in the Middle East and Africa can be a problem, due to the complex nature of regulatory approval and import processes. Unlike the EU, markets across MEA have their own rules and trade compliance regulations, presenting challenges for any international manufacturer.
Through its intimate knowledge of trade regulations, Pedigri Technologies can advise on import processes for each country, saving you the pain from doing so.
Indeed, it can successfully reduce products and spare parts import fulfillment lead time from an average 3-4 weeks to just one day or two.
This results in a shortened go-to market time for manufacturers, an improvement in field support SLAs for their hospital customers, and an avoidance of penalties and fines.
As its CEO Pramod Kattel explained:
"We are a specialised supply chain organization, or 5PL. We use services of various logistics integrators to move boxes between countries. But our trade compliance expertise, and unique IOR solutions offers the best in-class import solutions for manufacturers to optimise their working capital and improve their Field Support SLAs.
This is where we work with manufacturers to define regulatory and customs approval processes. Manufacturers from Europe, US or Asia for example, are unlikely to be familiar with Saudi customs law - and they don't need to be, as they can leave everything with us. We are the gatekeeper, handling trade compliance requirements, regulatory approvals, import clearance and last mile delivery to the hospital or clinic."
Today, Pedigri Technologies is supporting more than 45 global manufacturers in MEA with faster, more efficient, and regulatory compliant import processes.
As one customer said:
"We can now deliver our AI-based products very quickly, efficiently and with full transparency to our end customers in the Middle East and Africa region. Pedigri Technologies's demo management logistics solutions have helped us double our sales in the region thus helping us increase our local customer base and international presence."
Improved profitability through optimised inventory
A high in-country spare parts inventory holding at the end of the product life cycle means that many medical manufacturers write off millions of dollars worth of non-moving spare parts for equipment.
Pedigri Technologies offers advanced on-site VMI (Vendor Managed Inventory) management solutions that automate and integrate the process between manufacturers, distributors and hospitals, resulting in minimised discrepancies in on-site inventory and billing, and reduced inventory write-offs.
Through proprietary cloud-based PRODIGY software platform, for instance, Pedigri Technologies is able to collaborate with vendors to deliver the most efficient supply chain processes.
A leading Medical Equipment manufacturer commented:
"The successful partnership with Pedigri Technologies enabled us to achieve faster spare parts fulfilment (same/next business day) for our Medical Equipment warranty repairs. This improved our field support KPIs/ SLAs, customer satisfaction and our ROI. Their expertise in trade compliance and product registrations with regulators considerably reduced our new product's time to market."
Consider also Pedigri Technologies' cutting-edge Forecast as a Service (FaaS) solution, which optimises inventory and reduces obsolete spare part write-offs through the power of AI-enabled predictive analytics.
Through accurate forecasting algorithms the company can manage operations with 30-40% less inventory and is able to reduce up to 80- 90% end of life excess inventory.
By reducing in-country inventory levels by up to 40 %, furthermore, it can optimise working capital and improve profitability.
Cold supply chain logistics for sensitive products
Specialty cold chain logistics requires in-depth technology integration, along with logistics expertise, to transport sensitive goods in temperatures ranging from -20C to +25C. This is often lacking in 3PL organisations who might otherwise be familiar with product logistics.
Pedigri Technologies integrates cutting-edge package monitoring technologies on top of the 3PL layer to securely and quickly move sensitive products.
Its PRODIGY platform for example allows real-time monitoring of a package's location and storage temperature through a dashboard, any attempts to tamper, and Last Mile Delivery and real time view of Proof of Delivery.
It will also provide dry ice/cold pack refilling capability during transit, extended cut-off time for bio sample collection, and in-country consolidation points for smooth international shipping, among other benefits.

How Pedigri Technologies helped a leading global medical devices manufacturer
Despite working with local distributors, a top 3 global medical devices manufacturer in the UAE encountered a series of problems that included:
- Delays in order fulfilment on the part of local distributors
- Meeting Field Support SLAs
- Higher cost of product warranty management
- High working capital requirements due to high in-country inventory holding (itself due to longer shipment import lead times)
- Delays in obtaining regulatory approvals
- High end-of-life spare parts write-offs due to unplanned in-country inventory management
- Lack of real-time visibility on spare parts inventories and last mile delivery
Through its expertise in trade compliance and IOR services, Pedigri Technologies was able to achieve the following:
- Faster approval from regulatory authorities
- Reduced import fulfillment lead time from 3-4 weeks to 1-2 business days
- Substantially improved KPIs and field support SLAs for local market customers relating to warranty management and maintenance contracts
- Improved ROI for the vendor's customers by reducing the downtime of equipment due to the faster fulfilment of spare parts
- 30% percent reduction in working capital for the vendor due to a reduced local inventory holding
- End of life inventory write-off reduction from 30 % to 5 %, leading to improved ROI for the vendor
Speak to Pedigri Technologies today
If you are reading this and facing similar supply chain challenges in the Middle East and Africa today, don't hesitate to reach out to Pedigri Technologies, who will ensure compliance and faster import solutions to improve vendor sales, go-to-market time and field support SLAs.
Drop Pedigri Technologies an email at [email protected], or visit the company website at www.pedigritechnologies.com for more information.


